Interview with Dr. Claudio Polisseni about deep tissue neuronal research using Multiphoton Microsopy
Unlocking the Secrets of the Axolotl Brain: A study on the nervous system of salamanders reveals new insights on the evolution of limb movement and coordination. With remarkable regenerative abilities and under-explored neural circuits, scientists are developing cutting-edge methods to observe the functional connectivity of axolotl neurons.


Dr. Claudio Polisseni
Tanaka Lab
Research Institute of Molecular Pathology (IMP)

What is the focus of your research group?
Our research group, part of the Tanaka lab at the Research Institute of Molecular Pathology (IMP) in Vienna, is investigating and studying the nervous system of salamanders, in particular the ambystoma mexicanum, more commonly known as axolotl. These animals not only have remarkable regenerative abilities, including in the brain, but are also among the first animals during evolution to have developed a fully functioning set of limbs. Studying their brain can therefore shed light on the evolution of the nervous system and our ability to move arms and legs in a coordinated manner.
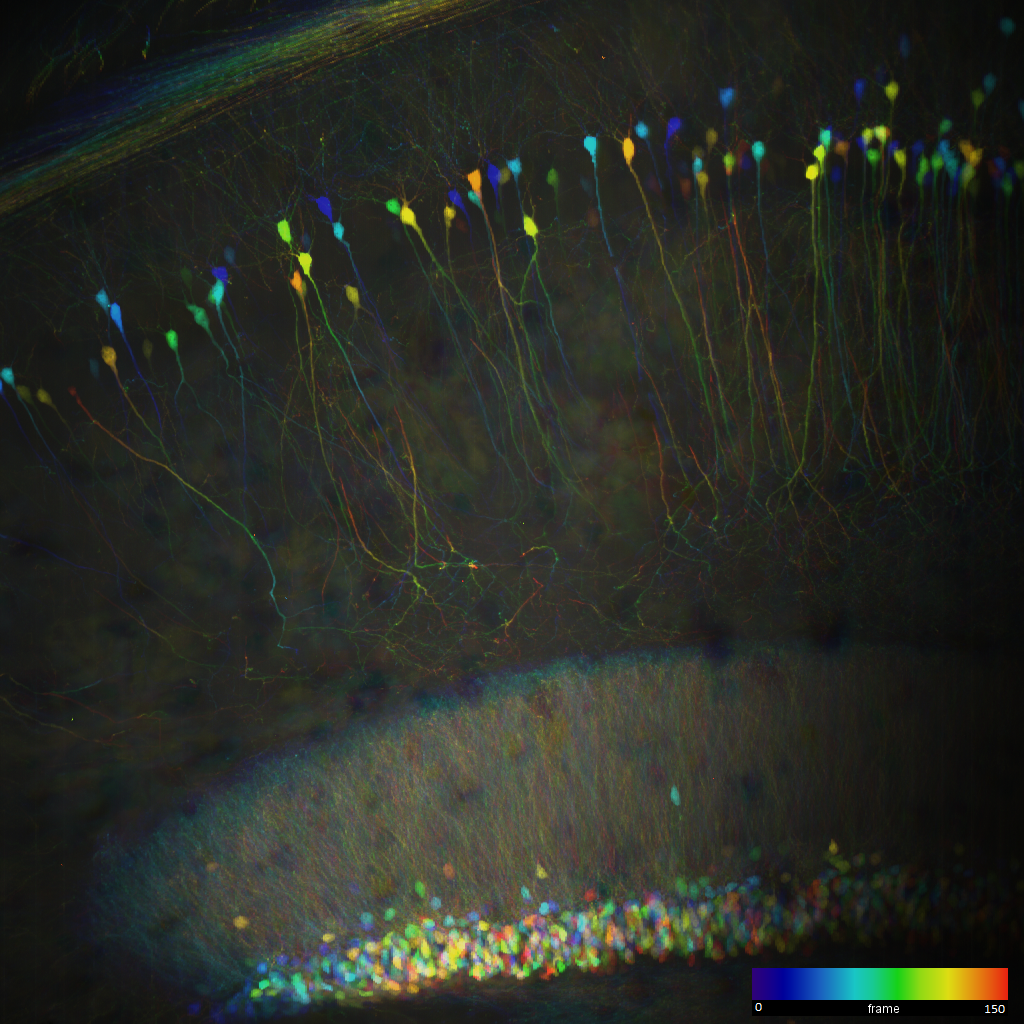
A depth-colour coded image of a z-stack through 150 um of an uncleared Thy1-GFP mouse brain
Can you go into detail on your experiments and what shall be achieved/improved?
While the nervous systems of zebrafish and rodents have been extensively studied, little is known about salamanders. We are currently developing methods to investigate the brain of these organisms. In particular, we are developing a method to reliably observe and measure the functional connectivity between neurons. We are integrating calcium indicators in the salamanders in order to then investigate different circuits, for example the olfactory and visual ones.
What are the critical points in the experiment and how does the Spark Lasers ALCOR help?
To investigate the functional connectivity of the axolotl brain and carry out in-vivo behavioral experiments in which neuronal activity is monitored upon stimuli, we have built an upright two-photon microscope. In fact, the nonlinearity of the two-photon absorption and the less scattering of near-infrared wavelengths, provide inherit 3D resolution and confined fluorescence in native tissues, such as the intact brain.
For our microscope we needed a very versatile, reliable, and high-power femtosecond laser source as little research has been done on the brain of salamanders and we expect the optical setup to be ever changing, introducing and integrating different upgrades. We aim at visualizing neuronal activity through GCaMP indicators, whose two-photon cross section peak is around 920 nm. Taking care of all these requirements, our system of choice has been the ALCOR-920-XSight-2W with integrated AOM.
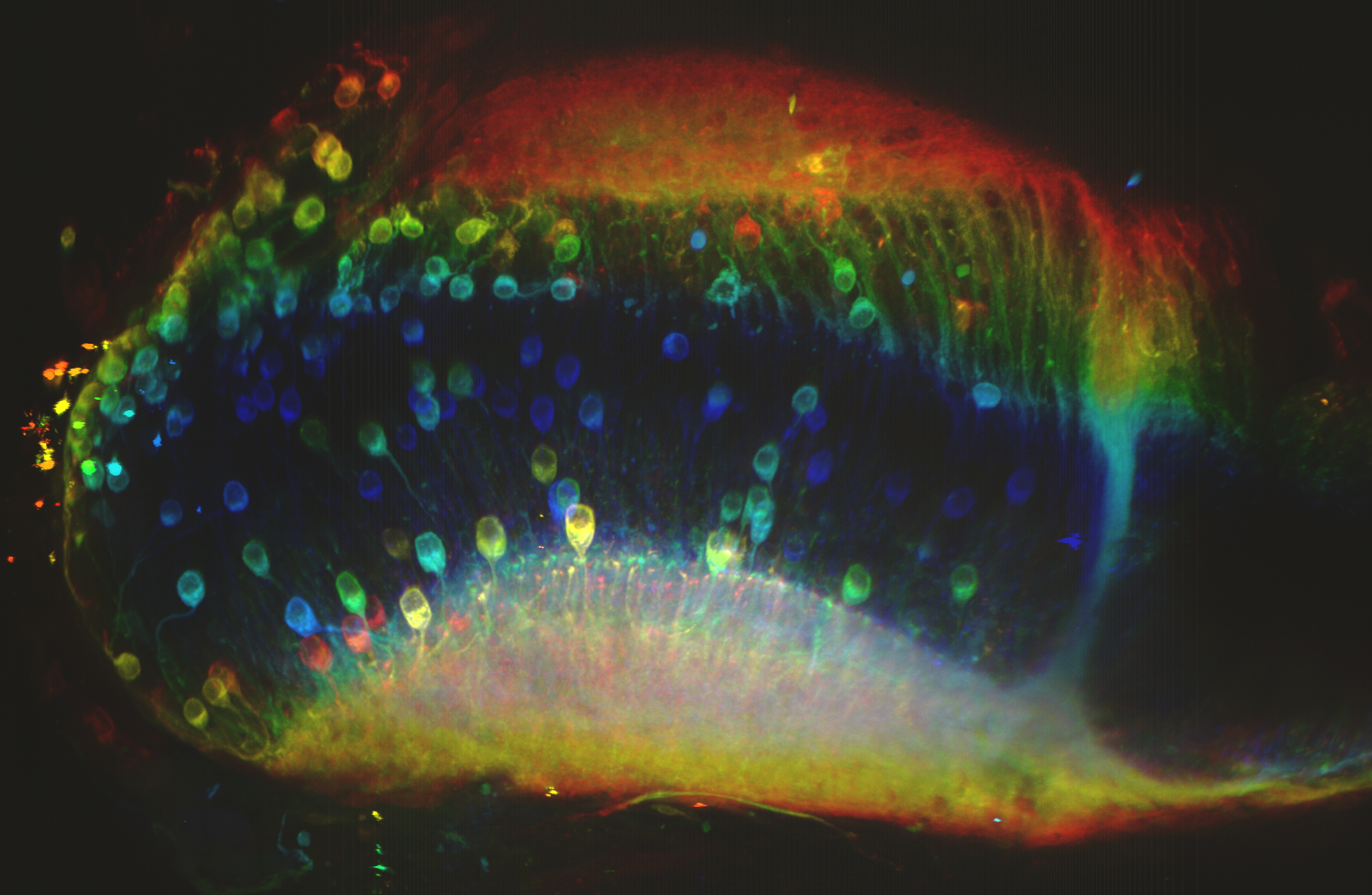
3D projection of cells in the optic tectum of a Beta-3 Tubulin – GFP transgenic axolotl. Stack size >400 µm.
Why did you choose Laser 2000 as a partner?
The ALCOR-920 provided the highest flexibility of any femtosecond laser source because, as I mentioned, we do not know what to expect looking into the brain of axolotls. As the laser light also has to travel through the thick layers of skin and skull, we required the highest possible power, confined into the shortest pulse of < 100 fs. This alone dramatically improves the signal-to-noise ratio. The computer-controlled Group Delay Dispersion (GDD) with a range of 0 to -60 000 fs² will also allow us to introduce new upgrades and more exotic optical components. In fact, after going through only a few lenses the GDD is now already set to -14 000 fs².
Did you face unexpected curveballs when implementing our product? If so, how did our team help you surmount those challenges?
I prepared for the arrival of the laser purchasing a few stands and mounts as well as beam samplers and photodiodes to measure and control the power output of the AOM. In the end, to my surprise, none of those components were necessary. The laser is plug-and-play and can be easily set and fixed at different heights and the AOM provides a linear output which is directly and easily controlled through the Spark Lasers software. In the end the only support we received was in choosing appropriate protective equipment and signs.
3D-movie of a hand of a Beta-3 Tubulin – GFP transgenic axolotl.
How do you envision the new version of the ALCOR 920 2.5 Watts would affect your results?
The optimized pulse quality as well as the higher power would definitely allow us to achieve better image quality imaging through the animals’ skins and skulls. It would also allow enough power to insert very lossy imaging components such as a remote focusing unit.
Thank you very much for your feedback and the constructive collaboration, Claudio!